Reconstitution reveals how myosin-VI self-organises to generate a dynamic mechanism of membrane sculpting
Benoit Rogez*, Laeschkir Würthner*, Anastasia Petrova, Dario Saczko-Brack, Maria-Ana Huergo, Christopher Batters, Erwin Frey and Claudia Veigel
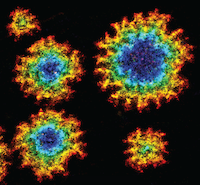
Active remodelling of the membrane curvature is key to cellular motile processes, including cell migration, endocytosis and polarisation in morphogenesis and development. A fundamental problem of molecular cell biology in conjunction with physics and mathematics is therefore to understand the evolutionary, developmental and functional rationale for these membrane shapes, as well as the mechanisms used by cells to produce them.
The sequence of events during membrane reshaping in various cell biological contexts remains controversial. According to the generally accepted view however, molecular motors get involved at the later stages of membrane trafficking, endocytosis, cell polarisation etc., while other, specialised proteins are responsible for initial local membrane reshaping. Our data strongly challenge this classical view and show that the motor proteins can get involved in the initial stages of membrane remodelling, and in the initial marking of sites for specific cellular functions, which dramatically changes the sequence of membrane remodelling events.
We report that myosin-VI on its own, remarkably, remodels the membrane to form rugged, flower-shaped membrane pores. We discovered that the completely unexpected, curvature-dependent and cooperative binding kinetics of the myosin favour saddle-shaped membrane geometries, which lead to characteristic and growing spatial patterns. Triangular-shaped gold nano-particles, combined with super-resolution microscopy, made it possible to reveal the preference for saddle-shaped geometries. We propose a quantitative theoretical model that describes this new route of protein-membrane interaction and the ensuing dynamics of membrane morphology, which we call a dynamic membrane sculpting mechanism.
The results challenge and broaden the classical view of the role of motor proteins in membrane sculpting by introducing a completely new function of motor – lipid interaction. The study also reveals the unexpected result that the motor lipid-binding does not depend on the presence of specific PIP(2) lipids, but instead on the shape of the bilayer, and that it is highly selective for saddle-shaped membrane geometries. Furthermore, binding occurs in almost equally spaced hotspots, observations hinting at a novel feedback mechanism between protein binding and membrane curvature.
We developed a theoretical model that correctly reproduces the key qualitative features of this protein-lipid interface, including flower-shaped morphology, memory effects for triangle-shaped pores, spontaneous emergence of new hotspots, and tip-splitting. Importantly, it also quantitatively explains the mechanism underlying patterning of equally spaced myosin-binding hotspots. Moreover, the theoretical concepts developed here may be broadly applicable to biological processes coupling morphology and biochemical processes.
In addition to the lipid properties that are inherent to a physiological membrane bilayer, and the currently known distinct mechanisms that allow proteins to sense, stabilize or generate high local membrane curvature, we believe that we have identified a new mechanism of dynamic membrane sculpting by (motor-) protein-lipid interaction. In addition to its potential in motor-protein linked drug targeting applications, and consistent with the universality of the already identified mechanisms of membrane reshaping in eukaryotic cells, we think it very likely that our new assay and modelling approach will help to uncover additional mechanisms underlying membrane shaping in the near future, and contribute towards revealing the universal role of membrane curvature in cellular functions.

