Limited Resources Induce Bistability in Microtubule Length Regulation
Matthias Rank, Aniruddha Mitra, Louis Reese, Stefan Diez, Erwin Frey
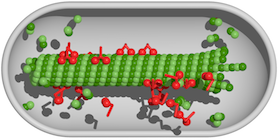 Molecular motors of the kinesin-8 family are important regulators of the length of microtubules, filamentous polymers which play vital roles in chromosome segregation and molecular transport. In our work we have addressed the question how length regulation functions in compartments of finite size, i.e., when the availability of protein is limited.
Molecular motors of the kinesin-8 family are important regulators of the length of microtubules, filamentous polymers which play vital roles in chromosome segregation and molecular transport. In our work we have addressed the question how length regulation functions in compartments of finite size, i.e., when the availability of protein is limited.
Cells possess an internal skeleton, which enables them to alter their form and actively migrate. This ‘cytoskeleton’ is composed of a number of filament systems, of which microtubules are one. As the name suggests, a microtubule is a cylinder. Its wall is made up of 13 protofilaments, each consisting of heterodimeric subunits containing two related tubulin proteins. Microtubules not only confer mechanical stability on cells and help to dictate their forms, they also serve as an intracellular transport network. Furthermore, microtubules are the major constituents of the mitotic spindle, which mediates the orderly segregation of the replicated chromosome sets into the two daughter cells during cell division. All of these functions require dynamic regulation of microtubule lengths. In our work we have, in collaboration with Professor Stefan Diez (Technical University of Dresden and Max Planck Institute for Molecular Cell Biology and Genetics, Dresden), developed and experimentally validated a model in which the motor proteins that are responsible for the transport of cargo along protofilaments also serve to regulate microtubule lengths.
The present study is based on earlier work conducted in our group [1], in which it was shown that the density of molecular motors attached to the filaments has an impact on whether the microtubule grows or shrinks, and that their effect depends on the length of the filament concerned. The longer the microtubule, the greater the number of motor proteins it can accommodate. Motor molecules of the family kinesin-8 proceed along the protofilament, stepping from one dimer to the next. When a kinesin protein reaches the end, it detaches from the filament taking the tubulin to which it is bound with it. Consequently, if the motor density on the protofilament is high, shrinkage will continue. On the other hand, a new tubulin dimer can bind to the end. At the end, motor-dependent shrinkage thus competes with microtubule growth. Assuming that resources (i.e. both tubulins and molecular motors) are abundant, there is a filament length at which the rates of growth and shrinkage balance out. However, in a real cell, these components are unlikely to be available in unlimited amounts. For example, formation of the mitotic spindle significantly depletes the numbers of free tubulin molecules in the soluble phase of the cytoplasm. In the new study, we explored the effects of such resource limitation on the regulation of microtubule length.
Using simulations based on a mathematical model of polymer dynamics, we found that under these conditions two distinct mechanisms of length regulation come into play. Which of these becomes dominant depends on the relative concentrations of the tubulins and the motor proteins: In a certain concentration range the dynamic equilibrium between growth and shrinkage of the microtubules operates as it would if resources were not limiting. A different mechanism becomes important when one of the required resources is in short supply. For instance, when not enough motor molecules are available to trigger rapid depolymerization of the protofilaments, the microtubules continue to grow until the concentration of tubulins falls below a critical value. Furthermore, there is a concentration range in which both processes are active. In this case, we observe that the microtubules come in two sizes and that they sometimes switch between the two lengths. This can be described as a phase transition. The theoretical predictions have been confirmed by in vitro experiments carried out by our collaborators in Dresden. In particular, our finding of a transitional bistable regime was shown to correspond to a range of concentrations in which microtubule lengths follow a bimodal distribution.
We are convinced that our results are also applicable to related systems. In particular, we suspect that the limitation of key resources may play an important part in regulating other cellular processes.
[1] Anna Melbinger, Louis Reese, and Erwin Frey, Phys. Rev. Lett. 108, 258104 (2012)
Related Links
- Phys. Rev. Lett. 120 (14), 148101 (2018): Limited Resources Induce Bistability in Microtubule Length Regulation
- LMU press release (German)
- LMU press release (English)

