Protein Recruitment through Indirect Mechanochemical Interactions
 Many key intracellular processes are spatially guided by self-organized protein patterns, which form through an interplay between diffusion and biochemical reactions. Biochemical coupling (via protein attachment) between cytosol and membrane is a crucial and recurring motif among the formation of protein patterns. In particular, some key proteins are capable of recruiting other proteins from the cytosol to the membrane, which results in a nonlinear feedback mechanism.
Many key intracellular processes are spatially guided by self-organized protein patterns, which form through an interplay between diffusion and biochemical reactions. Biochemical coupling (via protein attachment) between cytosol and membrane is a crucial and recurring motif among the formation of protein patterns. In particular, some key proteins are capable of recruiting other proteins from the cytosol to the membrane, which results in a nonlinear feedback mechanism.
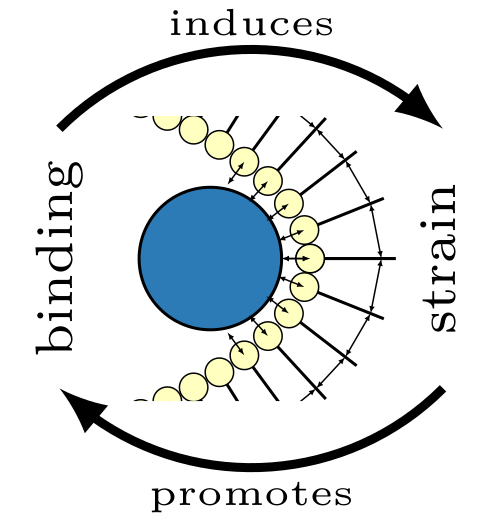
We propose a mechanistic picture for protein recruitment, which is motivated by well-studied membrane-bending properties of some protein species, like those containing BAR-domains. We devise a minimal model for membrane deformations induced by protein binding, based on the idea that the binding energy of a protein depends on the membrane conformation. Then, recruitment occurs through a competition between the energy gain for binding proteins and an energy cost for deforming the membrane. At low membrane-bound protein densities, binding is disfavored, leading to a low attachment and a high detachment rate. As more proteins attach to the membrane, the membrane state gradually becomes more binding-favorable, thus invoking nonlinear positive feedback in the attachment rate. These results are general and independent on the exact mechanism of protein binding and are equally applicable to membrane-bending and membrane-inserting proteins.
We predict that the nonlinearity of protein recruitment can be tuned by modification of protein-membrane interactions or by shifting the membrane rest state. Therefore, introducing permanently bound membrane inclusions (transmembrane proteins) might drastically change the protein patterns that are observed. Furthermore, since recent experiments by the Schwille group have shown MinD-induced deformations in giant unilamellar vesicles, we speculate that membrane-mediated recruitment is generally applicable to the broad class of NTPases that are essential for cellular protein pattern formation. Therefore, strain sensing and generation might be a prominent and perhaps general feature among membrane-binding proteins.

