Broken detailed balance of filament dynamics in active networks
J. Gladrow, N. Fakhri, F.C. MacKintosh, C.F. Schmidt, C.P. Broedersz
Living systems form an intriguing class of active soft matter. Indeed, cells are constantly fighting decay to thermal equilibrium, at which they would be dead. On the molecular scale, their non-equilibrium dynamics involves a complex and seemingly random arrangement of motions and forces. Due to this randomness, however, active jittering of nano-sized particles inside biological systems can be mistaken for Brownian motion. A natural non-invasive method to study how non-equilibrium dynamics manifests on different scales in active matter is by observing fluctuations of a tracer filament.
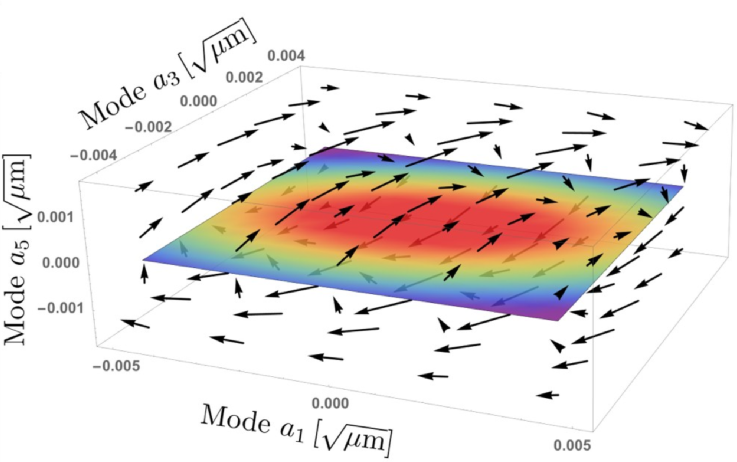
In our recent paper [1], we develop a theory for the non-equilibrium dynamics of such tracer filaments in a motor-activated network. Our model demonstrates how a breaking of detailed balance in the dynamics of embedded filaments can be harnessed to detect and measure activity in the surrounding network. We decompose the filament shape into Fourier m odes, to obtain a set of mode amplitudes, which captures the dynamics at a certain length scale. We find that these modes no longer evolve independently under nonequilibrium driving. This effective mode coupling results in nonzero circulatory currents in a conformational phase space, reflecting a violation of detailed balance. We present predictions for the characteristic frequencies associated with these currents and investigate how the temporal signatures of motor activity determine mode correlations, which we find to be consistent with recent experiments on microtubules embedded in cytoskeletal networks.

